Research by: Harrison Ndung’u Mwangi, Edward Kirwa Muge, Peter Waiganjo Wagacha, Albert Ndakala, Francis Jackim Mulaa
published in International Journal of Molecular Sciences here
Abstract
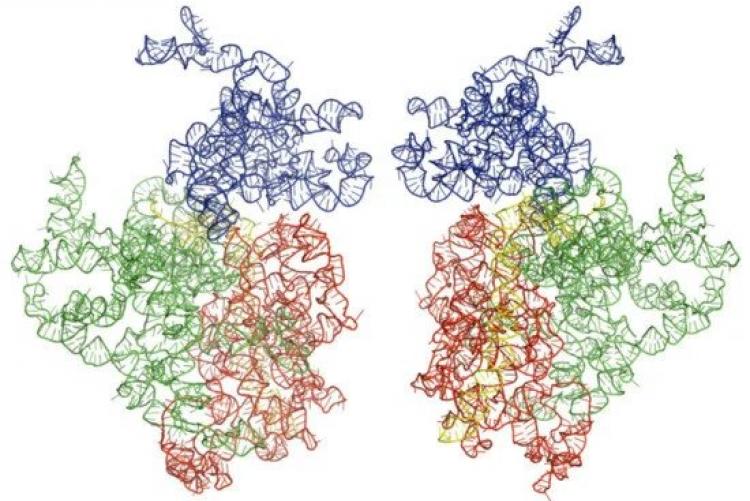
The development of novel anti-infectives against Kinetoplastids pathogens targeting proteins is a big problem occasioned by the antigenic variation in these parasites. This is also a global concern due to the zoonosis of these parasites, as they infect both humans and animals. Therefore, we need not only to create novel antibiotics, but also to speed up the development pipeline for these antibiotics. This may be achieved by using novel drug targets for Kinetoplastids drug discovery. In this study, we focused our attention on motifs of rRNA molecules that have been created using homology modeling. The RNA is the most ambiguous biopolymer in the kinetoplatid, which carries many different functions. For instance, tRNAs, rRNAs, and mRNAs are essential for gene expression both in the pro-and eukaryotes. However, all these types of RNAs have sequences with unique 3D structures that are specific for kinetoplastids only and can be used to shut down essential biochemical processes in kinetoplastids only. All these features make RNA very potent targets for antibacterial drug development. Here, we combine in silico methods combined with both computational biology and structure prediction tools to address our hypothesis. In this study, we outline a systematic approach for identifying kinetoplastid rRNA-ligand interactions and, more specifically, techniques that can be used to identify small molecules that target particular RNA. The high-resolution optimized model structures of these kineoplastids were generated using RNA 123, where all the stereochemical conflicts were solved and energies minimized to attain the best biological qualities. The high-resolution optimized model’s structures of these kinetoplastids were generated using RNA 123 where all the stereochemical conflicts were solved and energies minimized to attain the best biological qualities. These models were further analyzed to give their docking assessment reliability. Docking strategies, virtual screening, and fishing approaches successfully recognized novel and myriad macromolecular targets for the myxobacterial natural products with high binding affinities to exploit the unmet therapeutic needs. We demonstrate a sensible exploitation of virtual screening strategies to 18S rRNA using natural products interfaced with classical maximization of their efficacy in phamacognosy strategies that are well established. Integration of these virtual screening strategies in natural products chemistry and biochemistry research will spur the development of potential interventions to these tropical neglected diseases.
keywords: kinetoplastids; 18S rRNA; homology and de novo modeling; natural products; virtual screening; molecular docking
- Log in to post comments